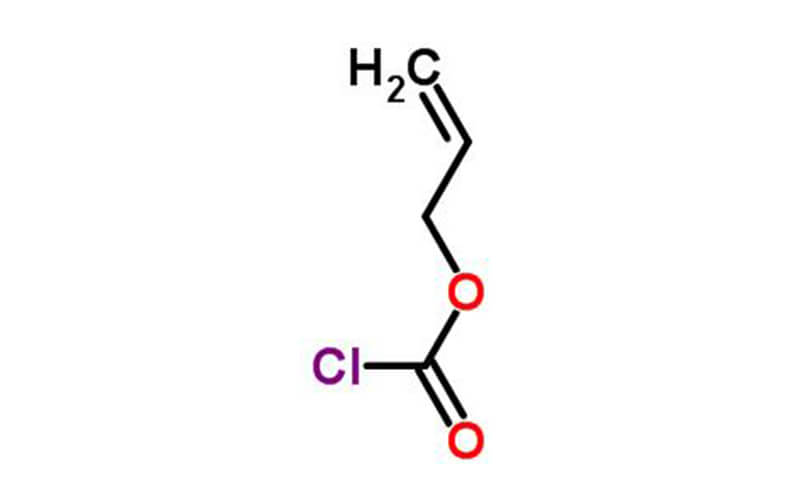
Allyl chloroformate Chemical Properties
| Melting point | 106℃ |
| Boiling point | 109 °C |
| density | 1.134 g/mL at 20 °C(lit.) |
| vapor pressure | 3.67 psi ( 20 °C) |
| refractive index | n20/D 1.422(lit.) |
| Fp | 88 °F |
| storage temp. | 2-8°C |
| solubility | Chloroform |
| form | Liquid |
| color | Clear slightly yellow to light orange to reddish brown |
| Odor | Extremely irritating, causes tears; pungent. |
| BRN | 773915 |
| Stability: | Moisture Sensitive |
Safety Information
| RIDADR | UN 1722 6.1/PG 1 |
| WGK Germany | 3 |
| RTECS | LQ5775000 |
| HazardClass | 6.1(b) |
| PackingGroup | I |
| HS Code | 29159000 |
| Hazardous Substances Data | 2937-50-0(Hazardous Substances Data) |
| Toxicity | mouse,LC50,inhalation,23100ug/m3 (23.1mg/m3),LUNGS, THORAX, OR RESPIRATION: DYSPNEA,Gigiena Truda i Professional’nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(5), Pg. 51, 1984. |
Allyl chloroformate Usage And Synthesis
| Chemical Properties | Colorless liquid |
| Uses | Allyl Chloroformate is used in the synthesis of poly(ethylene oxide) hybrid systems for therapeutic applications and drug delivery. Also used in the synthesis of benzothiazole derivatives with potent anti-tumor properties. |
| General Description | A colorless liquid with a pungent odor. Flash point 88°F. Corrosive to metals and tissue. Very toxic by inhalation, ingestion and/or skin contact. Vapors are heavier than air. |
| Air & Water Reactions | Highly flammable. Water reaction produces heat and acidic HCl. In the presence of moist air, corrosive hydrogen chloride is produced. |
| Reactivity Profile | Acid halides, such as Allyl chloroformate, are water reactive; some are violently reactive. They are incompatible with strong oxidizing agents, alcohols, amines, alkali. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291]. |
| Health Hazard | Vapor irritates eyes and respiratory tract. Contact with liquid causes eye and skin irritation, and ingestion irritates mouth and stomach. |
| Chemical Reactivity | Reactivity with Water Reacts slowly generating hydrogen chloride; Reactivity with Common Materials: Corrosive metals; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Flush with water, rinse with sodium bicarbonate solution; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent. |
| Safety Profile | Poison by inhalation and ingestion. Corrosive. Dangerous when exposed to heat, open flame (or sparks), or powerful oxidizers. Can react with oxidizing materials. To fight fire, use alcohol foam, spray or mist, dry chemical. When heated to decomposition it emits toxic fumes of Cl-. See also ALLYL COMPOUNDS and ESTERS. |
Our company specializes in hazardous chemicals, flammable and explosive chemicals, toxic chemicals (legal export), ultra-pure and high-purity reagents. Welcome to contact us.
Packing and shipping