How Ammonia Powers Nitric Acid Production: The Ostwald Process Explained
Nitric acid plays a key role in producing fertilizers, plastics, and explosives, making it indispensable in modern industries. Its production typically begins with ammonia, a compound that’s both abundant and affordable. The Ostwald process uses ammonia as a starting point, converting it into nitric acid through a series of catalytic and oxidation reactions. This method remains vital due to its efficiency and scalability, helping meet industrial demands worldwide.
Ostwald Process Overview
The Ostwald process is the backbone of nitric acid production, leveraging ammonia as its primary input. By converting ammonia into nitric acid efficiently, this method supports industries from fertilizers to explosives.
Historical Background: Development of the Ostwald Process and Its Impact on the Chemical Industry
The process was named after Wilhelm Ostwald, a German chemist who developed it in 1901. His innovation addressed the rising demand for nitric acid during a period when large-scale chemical production was becoming increasingly essential. By introducing a systematic catalytic oxidation system, Ostwald enabled the chemical industry to transition from older, less efficient methods to modern production techniques. Learn more about Wilhelm Ostwald and his contributions.
The process revolutionized the industry, making nitric acid widely accessible for manufacturing fertilizers like ammonium nitrate, synthetic fibers, and even explosives. Its introduction marked a shift toward higher efficiency and scalability in chemical production, laying the groundwork for today’s industrial operations.
Chemical Reactions Involved: From Ammonia to Nitric Acid
The Ostwald process hinges on three key reactions, each contributing to the conversion of ammonia (NH₃) into nitric acid (HNO₃):
- Oxidation of Ammonia:
- 4NH₃ + 5O₂ → 4NO + 6H₂O
- Ammonia is oxidized to nitric oxide (NO) in the presence of a platinum-rhodium catalyst.
- Oxidation of Nitric Oxide:
- 2NO + O₂ → 2NO₂
- Nitric oxide reacts with oxygen to form nitrogen dioxide (NO₂).
- Formation of Nitric Acid:
- 3NO₂ + H₂O → 2HNO₃ + NO
- Nitrogen dioxide dissolves in water, producing nitric acid and releasing additional nitric oxide to re-enter the cycle.
These steps exemplify a balance of efficiency and chemistry, ensuring minimal waste and maximum yield. Explore the reactions in-depth.
Catalysts Used: How Platinum and Rhodium Drive Efficiency
Catalysts are the unsung heroes of the Ostwald process. They speed up the oxidation of ammonia without being consumed in the reaction. A platinum-rhodium alloy is typically used, formed into fine gauze. Why this specific mixture? It offers unmatched efficiency and durability at high temperatures, resisting wear during repeated cycles.
Here’s how they work:
- Platinum: Provides stability and promotes the oxidation reaction.
- Rhodium: Strengthens the alloy, enhancing resistance to thermal degradation.
Using this alloy, the process achieves rapid and consistent results, even under intense operational conditions. If you’re curious about the mechanics of these catalysts, this resource dives deeper.
Process Efficiency: Balancing Yield and Energy Use
The Ostwald process achieves an impressive nitric acid yield of up to 98%. This high effectiveness arises from careful control of temperature, pressure, and reaction rates. However, it’s not entirely without drawbacks. Energy consumption remains substantial, with significant heat generated during the oxidation reactions. Cooling systems are essential to manage this energy while preventing unwanted side reactions.
Key factors influencing efficiency:
- Temperature Management: Reaction temperatures are maintained at roughly 900–950°C during oxidation.
- Energy Recovery: Excess heat is often recuperated to power other parts of the plant, reducing overall energy requirements.
Despite these challenges, ongoing innovations aim to reduce energy demands while maintaining the high output. Read about energy efficiency within the process.
The Ostwald process remains a cornerstone in the industrial production of nitric acid, striking a balance between chemistry, engineering, and sustainability.
Detailed Steps in Nitric Acid Production
Nitric acid production involves a series of precisely controlled chemical reactions. Each step is carefully orchestrated to maximize efficiency, minimize waste, and maintain safety. Below, we examine the key stages of this process, from ammonia oxidation to the absorption process.
Ammonia Oxidation
Ammonia oxidation is the initial and most critical step in the production of nitric acid. Ammonia (NH₃) is oxidized in the presence of a platinum-rhodium catalyst, which is finely gauzed to maximize surface area and reaction efficiency. This step creates nitrogen monoxide (NO) and water vapor.
Key parameters for this reaction:
- Temperature: Generally maintained at 900–950°C.
- Pressure: Typically operates between 4–10 atm.
- Reaction: 4NH₃ + 5O₂ → 4NO + 6H₂O
The high temperature and controlled oxygen levels ensure the optimal conversion of ammonia to nitrogen monoxide. The platinum-rhodium catalyst serves not just to speed up the reaction but also to withstand the intense conditions of this exothermic process. You can explore more about the catalysts and conditions for ammonia oxidation in this study on industrial processes.
Oxidation of Nitric Oxide
Once nitrogen monoxide (NO) is formed, it undergoes further oxidation to produce nitrogen dioxide (NO₂). This reaction occurs in the gas phase and is represented by the equation:
2NO + O₂ → 2NO₂
Key aspects of this step include:
- It takes place at atmospheric pressure to prevent unwanted side reactions.
- The reaction is spontaneous and exothermic, producing additional heat.
This step happens almost immediately as the NO molecules mix with oxygen in the air. Efficient gas mixing and the absence of contaminants are essential to ensure the rapid formation of NO₂. Detailed research on the kinetics of this phase, such as this analysis of NO oxidation, provides a deeper understanding of reaction speeds and catalyst impacts.
Absorption Process
The final step is the absorption of nitrogen dioxide (NO₂) into water to form nitric acid (HNO₃). This process typically occurs in a series of vertical absorption towers designed to maximize contact between NO₂ and water. During this step, lingering NO molecules are also recycled back into the system.
The chemical reaction is as follows: 3NO₂ + H₂O → 2HNO₃ + NO
Key considerations include:
- Temperature: Maintained at optimal levels to enhance absorption efficiency.
- Pressure: High enough to facilitate thorough mixing without excessive energy use.
During absorption, nitrogen dioxide reacts with water to form nitric acid. However, it also releases nitric oxide, which is recirculated back into earlier steps of the process — ensuring high overall efficiency. Studies like this research on NO₂ absorption reveal how process improvements have enhanced nitric acid yields over the years.
Each of these steps is vital for converting ammonia into high-yield nitric acid production. The precision involved ensures that the process is both cost-effective and sustainable.
Applications of Nitric Acid
Nitric acid is a vital component in many industries, driving advancements in agriculture, chemical synthesis, and beyond. Its versatile properties make it a cornerstone chemical with diverse applications. Below, we explore its major uses, delving into its role in fertilizer production, the manufacturing of other chemicals, and the environmental aspects surrounding its use.
Fertilizer Production: Detail how nitric acid is used in the manufacture of fertilizers
Fertilizers are perhaps the most prominent application of nitric acid. Agricultural productivity heavily relies on nitrogen-rich fertilizers, and nitric acid plays a crucial part in their manufacture. It reacts with ammonia to produce ammonium nitrate, a widely-used nitrogen fertilizer.
Key benefits of ammonium nitrate fertilizers:
- High nitrogen content enhances soil fertility.
- Boosts crop yields, ensuring food supply for growing populations.
- Easy to store and transport without losing effectiveness.
This use of nitric acid contributes significantly to addressing global food security challenges. According to Gasmet, modern nitric acid production processes are streamlined to meet the high demand for fertilizers while minimizing emissions.
Chemical Manufacturing: Explore other chemicals that are synthesized using nitric acid
Nitric acid is a key player in creating a range of other chemicals, acting as a chemical feedstock in numerous synthesis processes. Some significant uses include:
- Explosives Manufacturing: Nitroglycerin and trinitrotoluene (TNT) production utilize nitric acid, making it indispensable for both military and industrial explosives.
- Plastic Production: Serves in the synthesis of polyurethanes and other polymers.
- Dyes and Pigments: Nitric acid aids in producing nitro compounds used in vibrant dyes for textiles and paints.
Additionally, Lab Pro Inc. highlights its role as a cleaning agent in industrial processes. Nitric acid ensures the purity of equipment and maintains process integrity in high-precision industries like electronics and pharmaceuticals.
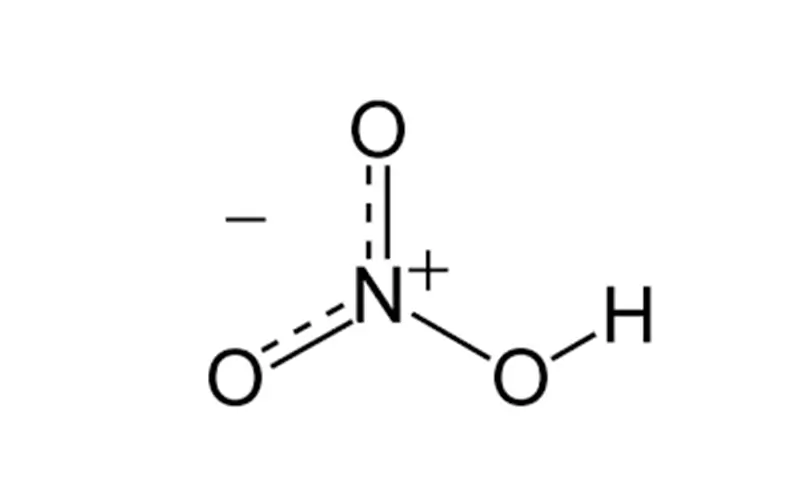
Environmental Considerations: Discuss the environmental impact of nitric acid production and the measures taken to mitigate it
The production of nitric acid is not without its environmental challenges. It releases nitrogen oxides (NOx) and nitrous oxide (N₂O), potent greenhouse gases that contribute to air pollution and climate change. However, modern processes have made strides in mitigating these effects.
Key environmental measures include:
- Catalytic Reduction Systems: These systems effectively lower NOx emissions during production.
- Efficient Plant Designs: Improved absorption towers and reaction conditions reduce waste gases.
- Recycling Ammonia Oxidation Byproducts: Minimizes raw material waste and emissions.
A report by the EPA discusses how advancements in nitric acid plants have drastically cut harmful emissions while maintaining high production yields.
Environmental consciousness in nitric acid production is essential not only for regulatory compliance but also for ensuring sustainability in the industries it serves. Continued innovation is crucial as producers balance industrial demand with ecological responsibility.
Future Trends in Nitric Acid Production
Nitric acid, a cornerstone of the chemical industry, is experiencing transformative changes amid evolving market demands and environmental priorities. These shifts signal emerging trends that could reshape its production in the future. In this section, we’ll explore two key factors driving these changes: sustainable practices and market dynamics.
Green Chemistry Innovations: Explore sustainable practices in nitric acid production
Sustainability is not just a buzzword—it’s becoming central to how nitric acid is produced. With increasing environmental scrutiny, manufacturers are now adopting greener technologies that minimize waste and emissions. But how exactly are they achieving this?
Here are several promising advancements:
- Low-Emission Technologies: Emerging methods focus on cutting down nitrous oxide (N₂O) emissions, a powerful greenhouse gas. Enhanced catalytic converters help reduce these pollutants significantly. Read more about these innovations.
- Alternative Feedstocks: Researchers are exploring synergies between sustainable ammonia (produced via green hydrogen) and nitric acid processes. This approach leverages renewable energy sources to reduce carbon footprints. Discover recent findings in sustainable feedstocks.
- Emission-Control Systems: Advanced plants now integrate stringent monitoring and control systems to keep emissions within minimal limits. Learn about emission reduction strategies.
These sustainable practices not only address regulatory requirements but also pave the way for a future where chemical processes align with global environmental goals. Imagine a future where nitric acid production is as clean as wind power—it’s not as far off as you think.
Potential Market Changes: Analyze market trends affecting the demand for nitric acid
The nitric acid market is evolving, shaped by industrial needs and global economic trends. It’s essential to understand how shifts in these areas influence its demand.
Here are some critical market factors to watch:
- Growing Fertilizer Use: As global populations increase, agriculture demands are rising, driving the need for ammonium nitrate fertilizers. This trend directly impacts nitric acid demand. Explore the market forecast.
- Rising Polymer Production: Nitric acid’s role in synthesizing plastics such as polyurethanes is bolstering its industrial importance. Learn more about market sectors contributing to growth.
- Shifting Geographic Demand: Emerging economies in Asia and South America are fueling higher consumption rates, with regional investments in industrial infrastructure. Check the regional analysis.
While demand remains strong, market volatility may arise from fluctuating raw material prices and stricter environmental regulations.







