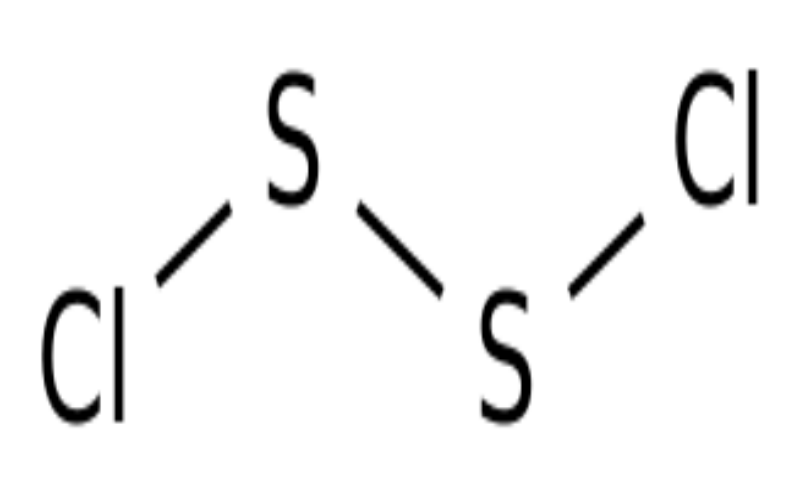Physical Properties
- Appearance: Amber to yellowish-red, oily, fuming liquid with a penetrating, suffocating odor.
- Density: 1.688 g/mL at 25°C.
- Melting Point: -80°C.
- Boiling Point: 138°C.
- Vapor Pressure: 6.8 mm Hg at 20°C.
- Refractive Index: n20/D 1.658.
- Solubility: Soluble in benzene, ether, carbon disulfide, and other organic solvents; reacts with water.
Chemical Properties
- Stability: Stable at room temperature, but decomposes to sulfur and chlorine at 100°C and completely at 300°C.
- Reactivity:
- Can be reduced by metals to form chlorides and sulfides.
- Reacts with chlorine gas to form sulfur dichloride.
- Reacts with metal oxides or sulfides to form metal chlorides.
- Decomposition: Forms sulfur, sulfur dioxide, and hydrochloric acid when hydrolyzed.
Uses
- Rubber Industry: Used as a low-temperature vulcanizing agent and adhesive.
- Organic Synthesis: Used as a chlorinating agent and for introducing C-S bonds.
- Metallurgy: Used as an extractant for precious metals like gold and silver.
- Other Applications: Used in the synthesis of lubricants, dyes, pesticides, and as a reagent in the Herz reaction.
Safety and Hazards
- Health Risks: Highly toxic; inhalation can cause severe respiratory irritation and lung edema.
- Corrosivity: Strongly corrosive to metals, especially in the presence of moisture.
- Storage: Store in a cool, dry place, away from moisture and incompatible materials.
Preparation
- Synthesis: Produced by passing chlorine gas into molten sulfur, followed by fractional distillation.
- Batch Process: Chlorine is bubbled into a solution of sulfur in S₂Cl₂ at 50-70°C until the desired chlorine content is reached.
- Continuous Process: Chlorine gas is passed into molten sulfur at 220-260°C, and the gaseous S₂Cl₂ is condensed.
Our company specializes in hazardous chemicals, flammable and explosive chemicals, toxic chemicals (legal export), ultra-pure and high-purity reagents. Welcome to contact us.
Packing and shipping