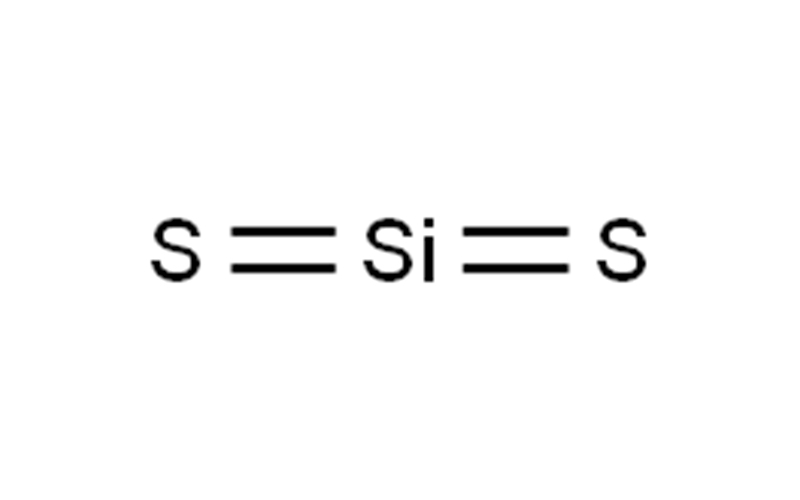Is Silicon Disulfide Ionic or Covalent?
Silicon disulfide (SiS₂) might not be a household name, but its chemical makeup raises an interesting question: is it ionic or covalent? As a compound where silicon bonds with sulfur, SiS₂ is held together by covalent bonds due to the nature of the elements . In this post, you’ll get a clearer picture of what defines its bonding properties and how its structure influences its behavior. Whether you’re tackling chemistry homework or just curious, this quick breakdown will provide the answers
Understanding Silicon Disulfide
Silicon disulfide (SiS₂) is a chemical compound that features intriguing properties, both structurally and physically. It shares some similarities with its more well-known cousin, silicon dioxide (SiO₂), but also houses distinct characteristics that set it apart. Dive into its molecular structure and physical traits to uncover why SiS₂ is noteworthy.
Chemical Structure of Silicon Disulfide
The molecular structure of silicon disulfide is linear, consisting of silicon atoms covalently bonded to sulfur atoms. Unlike silicon dioxide, which forms a robust three-dimensional lattice, SiS₂ adopts a chain-like structure of edge-sharing tetrahedra. This difference makes silicon disulfide less stable and more reactive compared to silicon dioxide.
In its chain structure, each silicon atom is bonded to two sulfur atoms, creating a repeating –Si(μ-S)₂Si(μ-S)₂– pattern. This configuration highlights the covalent nature of the bonds, which explains its non-conductivity and brittle nature. Unlike ionic compounds, where ions are held together by electrostatic forces, SiS₂’s molecular chains are bound by shared electron pairs, emphasizing its covalent identity.
Physical Properties
Silicon disulfide is best recognized for its pal white crystalline appearance. It is fragile and decomposes easily in the presence of water or alcohol, demonstrating its reactivity. Here are some key physical attributes:
- State at Room Temperature: Solid, resembling a fine white powder.
- Melting Point: Around 1,090°C, where it sublimates instead of melting directly. Reference
- Density: Approximately 1.85–2.02 g/cm³.
- Solubility: Decomposes in water yet is insoluble in non-polar solvents like benzene.
If you compare it with silicon dioxide, SiS₂ is significantly more reactive and fragile. SiO₂’s three-dimensional network makes it highly stable, whereas SiS₂’s chain-like structure leaves it prone to breakdown. Due to its sensitivity to water, silicon disulfide must be handled
For further details on its physical and chemical properties, you can check out this informative source. This comparison sets the stage for understanding its application and chemical behavior in different environments.
Ionic vs. Covalent Bonds
In the world of chemistry, understanding the difference between ionic and covalent bonds is key. These two types of bonds determine how atoms interact, how compounds behave, and why some substances are brittle while others are flexible. Let’s break down their unique characteristics to see how they differ.
Characteristics of Ionic Bonds
Ionic bonds are formed when one atom transfers electrons to another, creating charged ions that attract each other. This type of bonding is common between metals and non-metals. Here are the defining properties of ionic bonds:
- High Melting and Boiling Points: Ionic compounds require significant energy to break their strong electrostatic forces. See details on their properties here.
- Solubility in Water: These compounds dissolve in polar solvents like water, splitting into individual ions.
- Brittle Nature: Ionic compounds are hard yet prone to fracturing when stress is applied.
- Electrical Conductivity: When dissolved in water or melted, ionic compounds conduct electricity due to the free movement of ions.
- Crystal Structure: Ionic compounds often form well-organized, repeating lattice structures. Learn more about these properties here.
In essence, ionic bonds are like magnets—they stick together strongly under normal circumstances but can break apart under specific conditions.
Characteristics of Covalent Bonds
Covalent bonds occur when two atoms share electrons instead of transferring them. This sharing allows both atoms to achieve stability in their outer electron shells. Covalent bonding is typical between non-metal atoms. Take a look at the key features:
- Low Melting and Boiling Points: Covalent compounds are held together by weaker forces. Get deeper insights into these traits here.
- Poor Electrical Conductivity: Covalent compound do not conduct electricity, as they don’t have free-moving charged particles.
- Variety of States: Compounds can exist as solids, liquids, or gases at room temperature.
- Less Soluble in Water: Many covalent compounds don’t dissolve easily in water, especially nonpolar ones.
- Flexible Molecular Shapes: Covalent bonds allow for diverse structures, ranging from simple molecules to complex three-dimensional frameworks. Find additional details here.
Think of covalent bonds like a handshake—it’s a partnership where both parties share resources to maintain balance.
Silicon Bonds with Other Elements
Silicon, a versatile and abundant element, plays a critical role in countless chemical compounds. Its unique ability to bond with metals and nonmetals makes it a cornerstone of both biology and technology. The type of bonds silicon forms depends significantly on the elements it interacts with, leading to a diverse array of chemical behaviors.
Silicon with Metals vs. Nonmetals
When silicon bonds with metals, the interactions typically involve a transfer or sharing of electrons, lending unique traits to the resulting compounds. In these cases, silicon often behaves like a metalloid—a blend of metal and nonmetal properties.
- With Metals: Silicon can form metallic bonds or even engage in ionic bonds when paired with highly electropositive metals. A good example can be seen in silicon alloys, where its semi-metallic nature influences conductivity and thermal resistance. Learn more about these interactions here.
- With Nonmetals: Silicon’s interactions with nonmetals are predominantly covalent.Compounds like silicon dioxide (SiO₂) and silicon disulfide (SiS₂) showcase silicon’s ability to form strong, directional covalent bonds. Explore silicon’s covalent bonding tendencies further.
Silicon’s dual nature makes it a jack-of-all-trades in the chemical world, capable of creating robust frameworks or highly reactive complexes depending on its partners.
Bonding in Silicon Disulfide
Silicon disulfide (SiS₂) epitomizes the covalent bonding characteristics of silicon with nonmetals. Its structure features silicon atoms bonded to sulfur atoms in a linear, chain-like configuration. The bond types in SiS₂ are decidedly covalent for a few key reasons:
- Electron Sharing: Silicon and sulfur, both nonmetals, share electrons rather than transferring them. This shared bonding creates a stable yet directional molecular structure.
- Tetrahedral Geometry: Each silicon atom is surrounded by two sulfur atoms, adopting a repeating tetrahedral pattern. This arrangement mirrors the covalent chains seen in other silicon compounds like SiO₂ but lacks the lattice stability of a three-dimensional network.
- Lack of Ionic Properties: SiS₂ does not exhibit ionic behavior, such as electrical conductivity or high solubility in polar solvents. Its bonding is firmly within the realm of covalent chemistry. Find additional insights on SiS₂’s molecular structure here.
The covalent bonds in silicon disulfide contribute to its fragility and reactivity, especially when exposed to water or other polar environments. Its unique properties make it a fascinating study for chemists exploring silicon-based compounds.
Conclusion on Silicon Disulfide Bonding
Silicon disulfide (SiS₂) is a captivating example of how elemental properties define chemical behavior. Understanding its bonding nature is key to appreciating its characteristics and uses. SiS₂ is bonded covalently, showing how shared electron pairs create stability in its chain-like molecular structure. Let’s explore why this matters and its broader implications.
The Covalent Bonding in SiS₂
Covalent bonds drive the properties of silicon disulfide. Its structure is linear, with silicon atoms bonded to sulfur atoms in a repeating pattern. This covalent nature largely determines SiS₂’s behavior:
- Fragility: SiS₂’s chain-like arrangement lacks the rigidity of a lattice, making it more reactive and brittle.
- Reactivity: Exposure to water or alcohol decomposes silicon disulfide, showcasing the importance of its covalent interactions.
- Non-conductive Nature: Unlike ionic compounds, SiS₂ does not conduct electricity, highlighting the absence of free ions. Learn more about its physicochemical traits here.
Why Bonding Matters
The way silicon bonds with sulfur in SiS₂ sets it apart, influencing both its practical uses and chemical stability. Compared to ionic compounds, which have high melting points and electrical conductivity, SiS₂’s covalent bonds mean:
- It decomposes faster, requiring careful handling.
- It shows poor solubility in non-polar solvents, limiting certain applications.
- It stands out in specialized uses, like experimental materials in high-pressure environments. Explore application insights here.
Understanding these bonds isn’t just theoretical—it shapes how we approach silicon compounds in both academic research and industry. Silicon disulfide reflects the versatility of silicon, showing how one element can yield entirely different behaviors depending on its atomic companions.