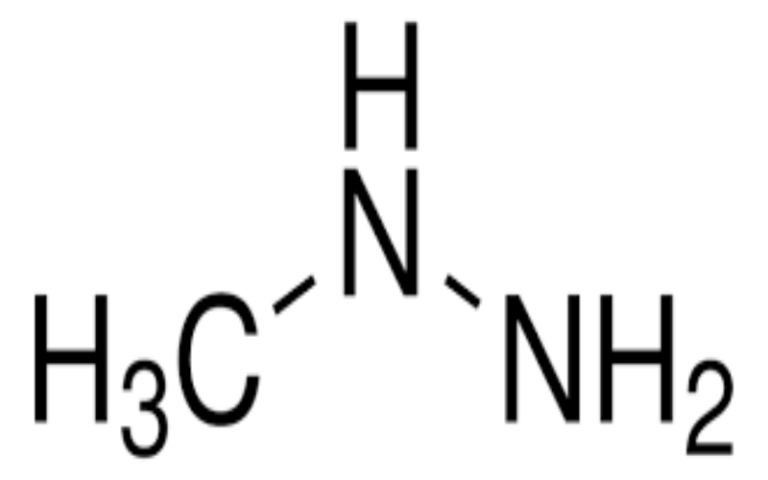
Physical Properties
- Appearance: Colorless gas with a fishy or ammonia-like odor.
- Boiling Point: -6.3°C.
- Melting Point: -93.5°C.
- Density: 0.7 g/mL (liquid).
- Solubility: Highly soluble in water, alcohol, and ether.
- Odor Threshold: 3.2 ppm.
- Vapor Pressure: 3 atm.
- Autoignition Temperature: 430°C.
- Explosive Limits: LEL = 4.9%, UEL = 20.7%.
Chemical Properties
- Basicity: Weakly alkaline, stronger than ammonia.
- Reactivity: Forms salts with acids, reacts with acid halides and anhydrides to form N-substituted amines.
Uses
- Organic Synthesis: Used as an intermediate for pharmaceuticals, pesticides, and surfactants.
- Industrial Applications: Used in the synthesis of dyes, rubber vulcanization accelerators, and ion-exchange resins.
- Other Uses: As a fuel additive, rocket propellant, and in leather tanning processes.
Production Methods
- Industrial Synthesis: Produced by the catalytic amination of methanol and ammonia at high temperature and pressure.
- Alternative Method: Can also be synthesized by reacting formaldehyde and ammonium chloride in the presence of zinc chloride.
Safety Information
- Toxicity: Inhalation toxicity is low; the maximum allowable air concentration is 5 mg/m³ (0.4 ppm).
- Health Hazards: Causes irritation to eyes, skin, and mucous membranes. Inhalation at high concentrations can cause severe respiratory damage.
- Fire Hazard: Highly flammable gas; forms explosive mixtures with air.
Storage and Handling
- Store in a cool, well-ventilated area, away from sources of ignition and oxidizing substances.
- Handle with care to avoid leaks and inhalation.
First Aid
- Skin Contact: Wash with water and apply acetic acid or citric acid solution.
- Eye Contact: Rinse with water for at least 15 minutes and seek medical attention.
- Inhalation: Move to fresh air and provide oxygen if breathing is difficult.
Our company specializes in hazardous chemicals, flammable and explosive chemicals, toxic chemicals (legal export), ultra-pure and high-purity reagents. Welcome to contact us.
Packing and shipping