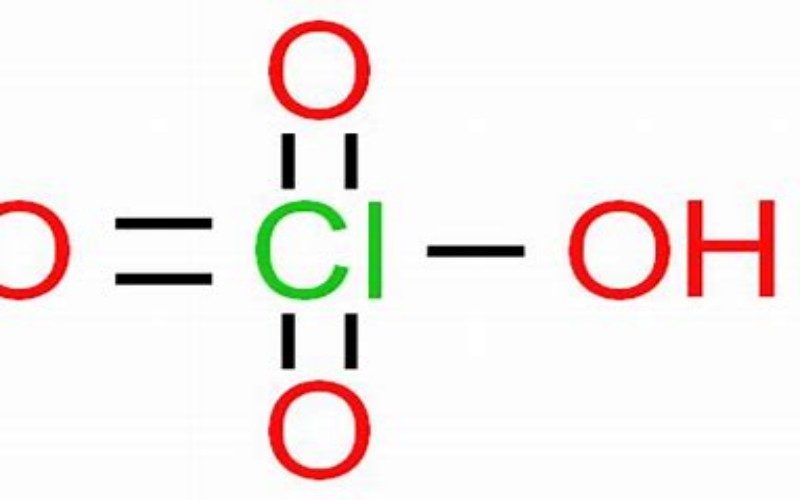
Physical Properties
- Appearance: Colorless, fuming liquid
- Odor: Odorless
- Melting Point: -18°C (for the azeotrope); -112°C (anhydrous)
- Boiling Point: 203°C (for the azeotrope)
- Density: 1.664 g/mL (at 25°C)
- Solubility: Soluble in water
- Vapor Pressure: 6.8 mm Hg (at 25°C)
- Refractive Index: 1.419
Chemical Properties
- Strong Oxidizing Agent: Perchloric acid is a powerful oxidizer and can react violently with reducing agents, potentially causing explosions.
- Strong Acidity: It is one of the strongest inorganic oxyacids and can react with most metals to form perchlorates.
- Decomposition: Anhydrous perchloric acid can decompose at high temperatures or in the presence of reducing agents, releasing chlorine and oxygen gases, which can be explosive.
- Stability: Relatively stable at room temperature but should be kept away from organic materials and reducing agents.
Preparation Methods
- Electrolysis: Electrolyzing sodium chlorate (NaClO₃) to produce perchloric acid.
- Double Decomposition: Reacting sodium perchlorate with concentrated sulfuric acid to generate perchloric acid.
Uses
- Laboratory Reagent: Used in analytical chemistry for acid digestion and titration.
- Industrial Applications: Used in the production of perchlorate salts, explosives, and rocket propellants.
- Electroplating: Used in electroplating processes for metals like gold and silver.
Safety Considerations
- Handling: Should be handled with extreme caution due to its strong oxidizing and corrosive properties.
- Storage: Store in a cool, well-ventilated area, away from organic materials and reducing agents.
- Emergency Measures: In case of spills, neutralize with a suitable base and clean up according to safety protocols.
Health and Environmental Hazards
- Toxicity: Highly corrosive and can cause severe burns to skin and eyes. Inhalation of vapors can cause respiratory irritation.
- Environmental Impact: Can be harmful to aquatic life and should be disposed of properly to avoid environmental contamination.
Our company specializes in hazardous chemicals, flammable and explosive chemicals, toxic chemicals (legal export), ultra-pure and high-purity reagents. Welcome to contact us.
Packing and shipping