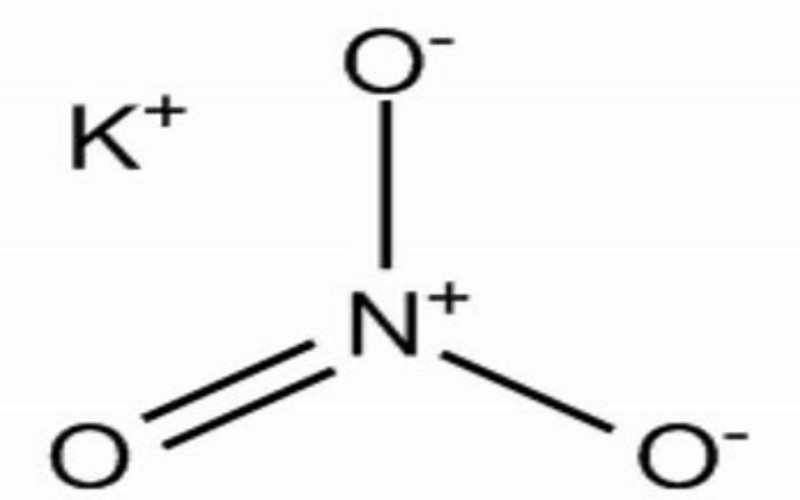
Physical Properties
- Appearance: Colorless transparent crystals or white granular powder.
- Density: 2.11 g/cm³ at 20°C.
- Melting Point: 334°C.
- Boiling Point: Decomposes at 400°C, releasing oxygen.
- Solubility:
- Soluble in water: 13.3 g/100 mL at 0°C, 247 g/100 mL at 100°C.
- Slightly soluble in ethanol, soluble in glycerol and liquid ammonia.
Chemical Properties
- Oxidizing Agent: Potassium nitrate is a strong oxidizer and can react violently with reducing agents, organic materials, and powdered metals.
- Stability: Stable at room temperature but decomposes at high temperatures, releasing toxic nitrogen oxides (NOₓ).
Uses
- Agriculture: Used as a fertilizer, providing both nitrogen and potassium (13% N, 46% K₂O) without chloride.
- Pyrotechnics and Explosives: Key component in gunpowder, fireworks, and rocket propellants.
- Food Industry: Used as a preservative and color fixative in meats.
- Medical Uses: Historically used as a diuretic and to treat hypertension; currently used in toothpastes to reduce tooth sensitivity.
- Other Industrial Uses: Used in glass manufacturing, matches, and as a flux in metallurgy.
Safety and Hazards
- Health Hazards: Inhalation or ingestion may cause irritation to the eyes, nose, and throat. Chronic exposure can lead to anemia and methemoglobinemia.
- Fire and Explosion Risk: Potassium nitrate is a powerful oxidizer and can accelerate combustion. It poses a significant fire and explosion risk when mixed with organic materials or reducing agents.
- Storage and Handling: Store in a cool, dry place away from organic materials and reducing agents. Use protective equipment when handling.
Environmental Considerations
- Ecotoxicity: Limited data available, but it is generally considered harmful to aquatic life.
- Disposal: Dispose of in accordance with local regulations to avoid environmental contamination.
Production Methods
- Commercial Production: Potassium nitrate is typically produced by reacting potassium chloride with nitric acid at high temperatures.
- Alternative Method: It can also be prepared by mixing hot saturated solutions of potassium chloride and sodium nitrate, followed by crystallization.
Our company specializes in hazardous chemicals, flammable and explosive chemicals, toxic chemicals (legal export), ultra-pure and high-purity reagents. Welcome to contact us.
Packing and shipping