Properties
| grade | ACS reagent |
| Quality Level | 200 |
| Assay | ≥99.0% |
| form | crystals, granules or powder |
| reaction suitability | reagent type: oxidant |
| impurities | ≤0.005% N compounds≤0.2% insolubles |
| pH | 7.2-9.7 (20 °C, 20 g/L) |
| anion traces | chloride, chlorate (as Cl-): ≤0.005%sulfate (SO42-): ≤0.02% |
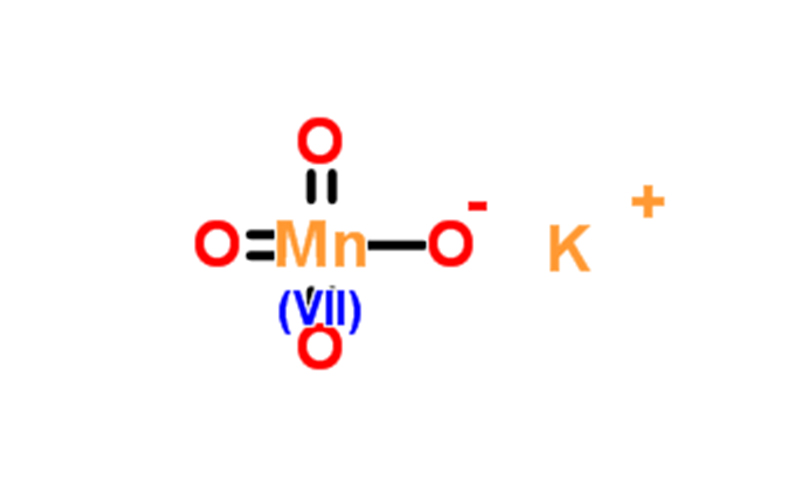
| SMILES string | [K+].[O-][Mn](=O)(=O)=O |
| InChI | 1S/K.Mn.4O/q+1;;;;;-1 |
| InChI key | VZJVWSHVAAUDKD-UHFFFAOYSA-N |
Application areas
- As an oxidant, it is used to make saccharin, vitamin C, isoniazid and benzoic acid, etc.
- In the environmental protection field, it is used as a water treatment agent to purify water and treat wastewater.
- In the metallurgical field, it is used to separate molybdenum from copper, remove impurities from zinc and cadmium, and float compounds.
- Bleaching agent for special fabrics, waxes, greases and resins, adsorbent for gas masks, and colorant for wood and copper.
Basic Information
- Chemical properties: Black purple, slender angular, granular, needle like or quicksand like crystals, with a blue metallic luster, odorless, sweet and astringent taste, sensitive to light. heated to about 240 ℃, decomposed with oxygen escaping, and decomposed when encountering ethanol and other organic solvents; It can also free oxygen from concentrated acid, and when it comes into contact with hydrochloric acid and free chlorine, it can be decomposed by most reducing substances, such as ferrous salts, iodides, and oxalates, especially when acid is present, it is more prone to oxidation. Easy to dissolve in boiling water, soluble in water. PH 7-9 (20g/L, H2O, 20 ℃). Melting point>240 ℃ (dec.). Density ρ (20) 2.70g/mL.
- Usage: Oxidizer; disinfectant; Chemical synthesis; Clean water; Reagents for capacity analysis oxidation-reduction method determination; Reagents for chromatographic analysis; Used for emission spectroscopy analysis; Drip analysis of barium, silver, sulfate, iron, vanadium, and tin, clinical examination of exfoliated cells, etc.
- Storage: Ordinary temperature
- Hazard statement: Combustion supporting, harmful, oxidizing and corrosive. Highly hazardous to water bodies.
- Preventive statement: Keep away from heat sources/hot surfaces/sparks/open flames/other ignition sources. Do not inhale. Wear protective clothing/gloves/goggles/masks. Avoid releasing into the environment. Firefighting: Misty water, carbon dioxide. Avoid taboo items.
Our company specializes in hazardous chemicals, flammable and explosive chemicals, toxic chemicals (legal export), ultra-pure and high-purity reagents. Welcome to contact us.
Packing and shipping